Alibaba Group cut net carbon emissions from its direct operations by 12.9% over its fiscal year, according to the company’s annual Environmental, Social and Governance (ESG) report published on Monday. The Hangzhou-based company is tapping…
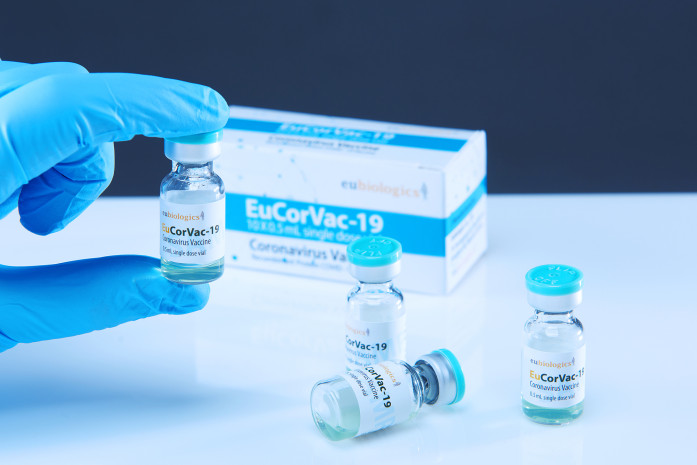
POP Biotechnologies (POP BIO), a Buffalo, NY-based biopharmaceutical startup, announces top line interim results of a Phase 3 clinical trial of EuCorVac-19, a COVID-19 vaccine candidate being developed by South Korean partner EuBiologics (KOSDAQ: 206650). EuCorVac-19…
Griffith Foods is proud to announce its near-term science-based emissions reduction targets have been validated by the Science Based Targets initiative (SBTi), a collaboration between CDP, the United Nations Global Compact, World Resources Institute (WRI)…
Nutritious eating, including smarter snacking, is an important way to protect your heart and maintain overall health and wellness. While many people understand the basics of healthy eating – avoiding too many sugary treats, for…
Lenovo signed a new agreement with Al Hathboor Bikal.ai, to provide public and private sector customers with cutting-edge high performance cloud computing systems to support advanced digital transformation projects which focus on developing innovation in…
In today’s fast-paced world, the rise of dad-bods among men is becoming increasingly prevalent. As we navigate the complexities of daily life, it’s all too easy for healthy habits to slip away. Juggling work, family,…
Mendocino Coast Healthcare Foundation announces the return of Winesong. The charity event will be held at the beautiful Mendocino Coast Botanical gardens on Saturday, Sept. 9, at 11:00 AM. This event is a spectacular combination…
Bringing consumerism to ePrescribing encourages patient adherence to drug therapies and stronger engagement in their health through timely and easy-to-understand pricing information. The RxWallet® application from RxEOB, a transformational health information technology company, offers patients…
While the company will also provide life coaching services, Dr. Grimes emphasizes that the main focus of Elevated Minds remains psychotherapy, and the expansion of life coaching will be an additional wellness service. The COVID-19…
Thanks to inquiring minds, 34,000 fewer young bellies were impacted by empty cupboards and empty stomachs in 2022. I Can for Kids Foundation (iCAN) was formed in 2015 after a television segment on student hunger…